Idea
I plan for my interaction with Physarum polychefalum to manifest in a sculptural habitat that connects the organism's natural habitat - the woods - with the life it has been habituated to lead in sterilised, monitored and controlled human captivity. To this end, I am visually comparing the structure of tree trunks and/or branches to the human body in a 'shape/form-studie'. The sculpture should invoke a corporeal impression: In this context the human body will represent an analogy for the human-centred ontological terminology commonly used in both the media and scientific representation and study around the unicellular organism. Terminology like 'intelligence', 'memory', 'social behaviour', which we are quickly invited to use in our understanding of this other organism. I want to question such human-centred categorisation and how this (subconsciously) shapes our encounters with such species.
To develop this sculpture I will follow two guiding questions:
How can I shape the parameters and variables of the habitat to realise my vision, while tending to the 'needs' of a living organism? (Needs: moisture - Agar; micro-organisms - Oatflakes) (Parameters: Light, temperature, humidity, aerosole, bacteria, altitude, etc.) I.e. Which parameters need to be closer to a lab context vs. which should more closely resemble the natural environment to ensure the vital (yellow, branching out) appearance?
How does my human influence manifest on a controlled, repeatable visual level? i.e. can I control the look (structure, color, movement)?
Technical description
When looking at the tree-habitat of PP the three main variables: moisture, 'food' and space appear invisible to our view. In the petri-dish, these variables are the most visible and limiting. So much so that they obstruct the visibility of the natural structure, colour and movement of the organism. I want to close the distance between these different conditions and open up a third environment: My sculpture will represent a meeting point between the natural and 'human-controlled' habitat. As such I will habituate PP from the Lab-context back towards more natural conditions in a process of adapting the 'living conditions/variables' between the varying degrees of synthetic to natural.
The parameters to be defined and controlled:
(1) Moisture - Agar The Biolab standard is a 100ml distilled water x 2g Agar solution. The natural condition is moist tree trunks and soil (moisture naked to the human eye).
Approach (1) Provide moisture by simulating rain and spraying the organism with (distilled) water. Approach (2) Create Agar/moisture 'pods' to show PP (at least partially) unobstructed. Akin to the idea of a watering hole in the desert: PP could draw its moisture from one spot from which it would branch out further possibly connecting these different 'moisture pods'.
(2) 'Food' - microorganisms, bacteria, fungi, yeast - oatflakes The Biolab standard are oatflakes, from which the slime mold 'eats' only the bacteria on the flakes. The natural condition are bacteria, yeasts, and fungi found on deciduous tree logs/wood drawn from the ground surface.
Approach (1) Investigate the question: How long can you live without me? by retreating the human influence and no longer 'feeding' it. Simultaneously, accepting that by releasing this control over the life cycle the slime mold might develop to spores or dry out, hence the artist looses control over the appearance. Approach (2) Subsidies the oatflakes to something actually found in nature and supply this 'food' over the ground surface i.e. Agar base. I.e. develop an Agar mix that is both providing the essential 'food' source while not obstructing the visual appearance.
It is important to note that all decisions on technical parameter are made primarily on a visual aesthetics basis. This means that my aim is to create the most unobstructed presentation of the slime mold as possible within my sculptural vision.
(3) Space + Surface
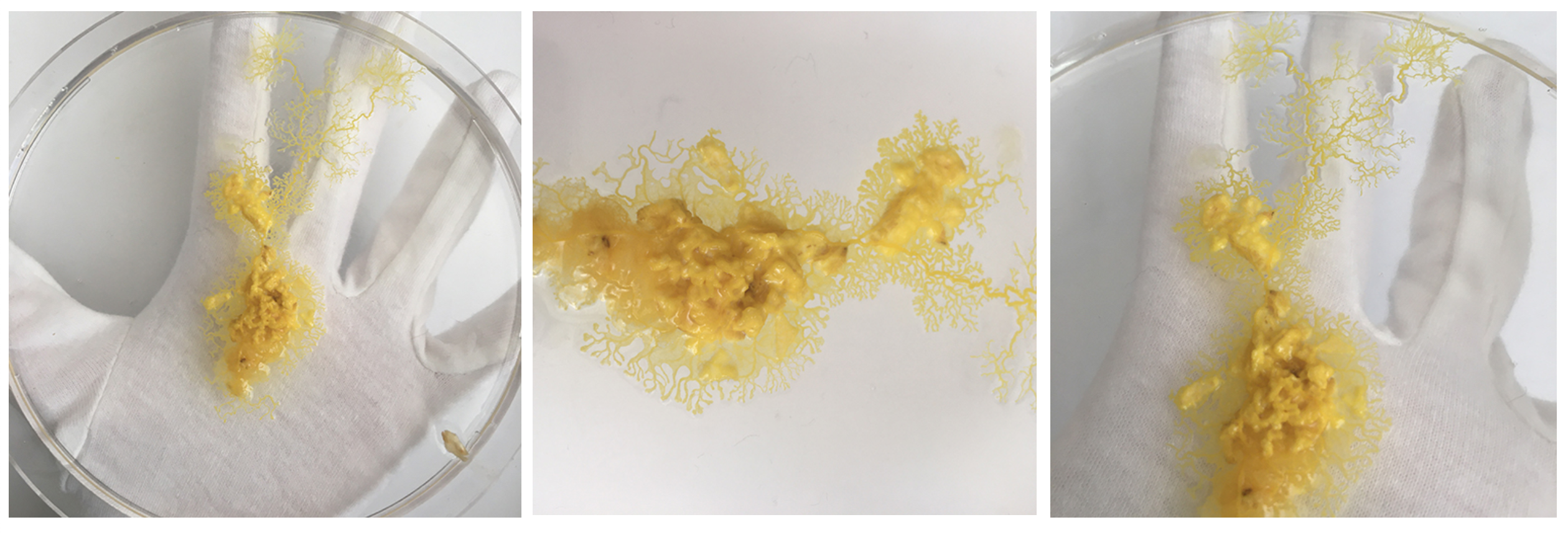
When looking at the tree-habitat the first thing that strikes me is the surface. The surface is an obstacle course over which the organism sustains its life. This vital parameter is all but omitted in the lab context, where is sterilised, small and made conform for scientific observation. This parameter - Space + Surface - is the core of my work.
Approach (1) Source/collect tree trunks that have a visual semblance to the human body. Develop this found artefact further into a sculptural object through casting techniques (possibly with wax). In a second step: test PP's survival capacities on sculptural surface.
In parallel: Approach (1.1) Condition PP to live in a larger space (67x37x25 glass box), lifting the restrictive spatial boundaries and allowing for more natural movement patterns to emerge.
(4) Additional parameter through human interaction: Care
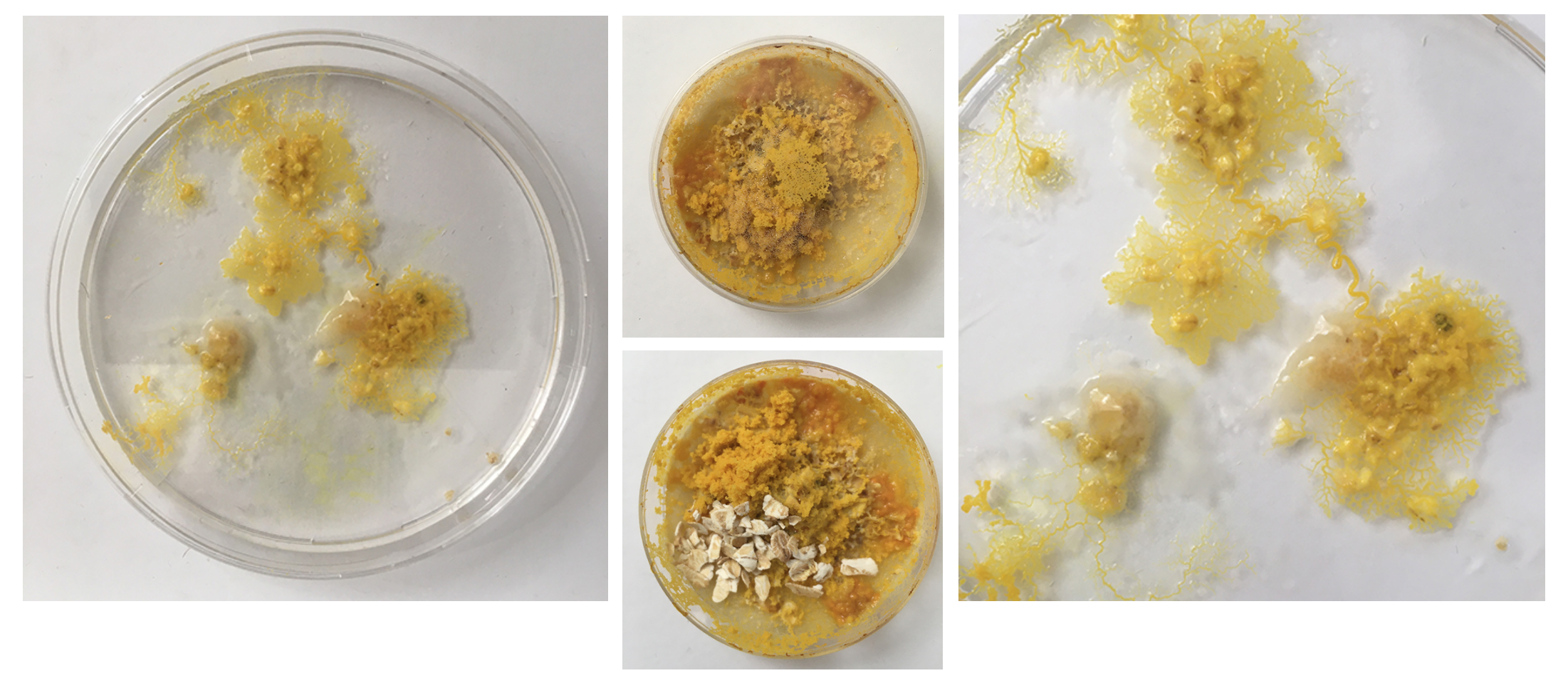
The method of inoculation can influence the appearance of Physarum polychefalum: Approach (1) Inoculate 'fresh' PP with a chunk of the oat flakes. Approach (2) Inoculate 'fresh' PP by scraping from the top of Physarum growing on oats. Approach (3) Inoculate 'medium fresh' PP by scraping the veins that are reaching towards the outer walls of the petri dish Approach (4) Inoculate 'old' PP by using the orange-y parts that have grown out of the dish.
Via recreating the same conditions several times and continued observation of these different methods of inoculation my understanding and control over the possibly visual structures and colourations of the organism grows.
This investigates the question to which degree my human influence can manifest on a repeatable visual level within the petri-dish environment. In a second step the control over the appearance will need to be transplanted to the sculpture.
Reflections and thoughts weeks 1 - 4
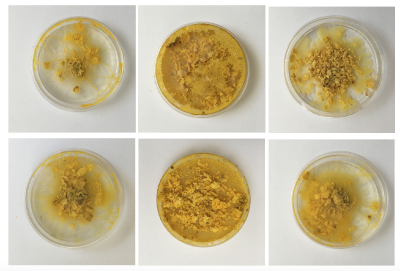
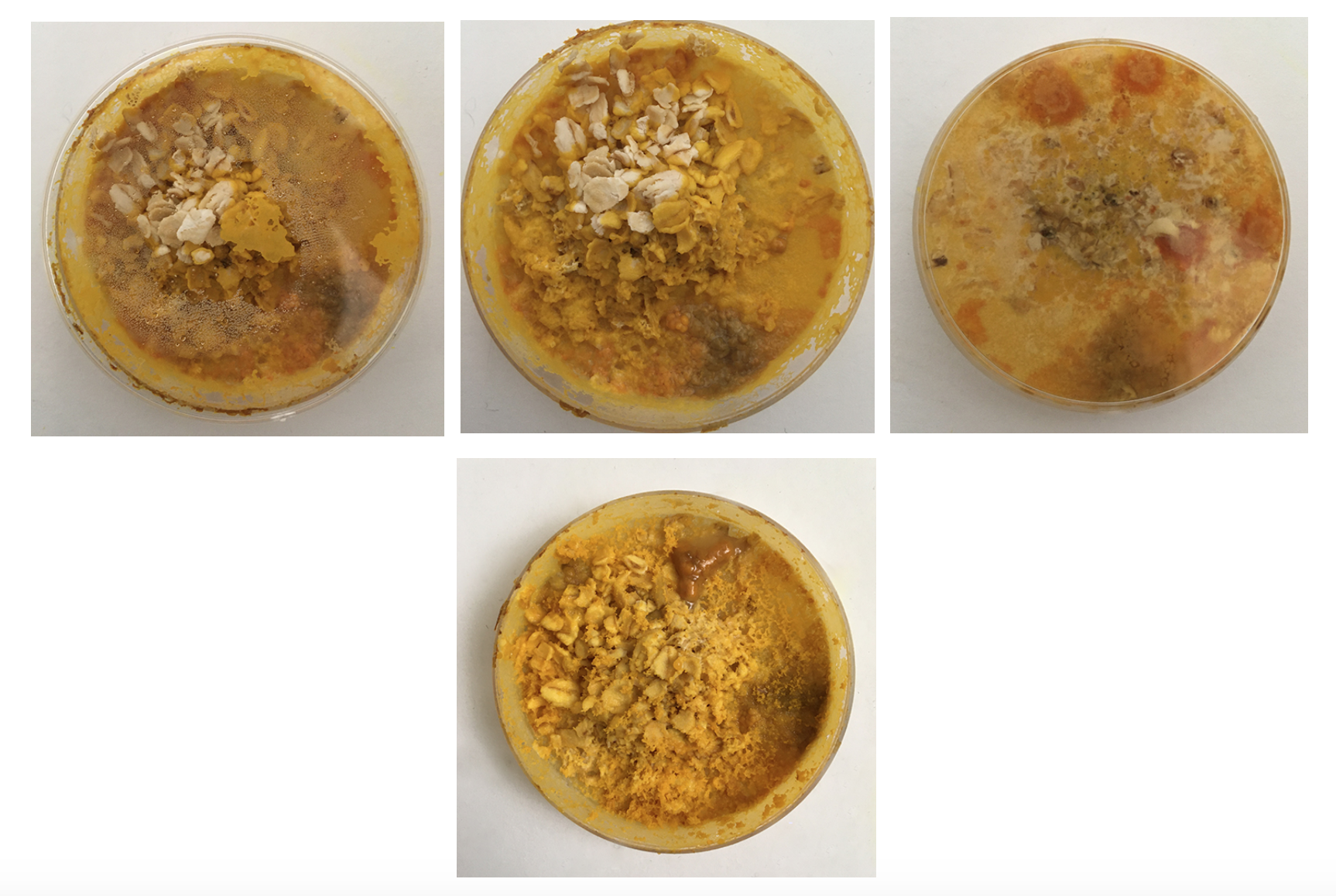
I have been documenting in the form of photographs a lot, simply because the organism is constantly evolving so that if not captured the moment will have passed. (As initially everything was new I was taking photographs of all dishes. I have since gone over to only documenting special structures, or bacterial infections or other unusual features.)
At the same time my point of view was never entirely free as I entered this project with (1) already an interest for (especially) the dynamic, mind-of-it's-own quality of a growing organism (also relates to Fungi) - meaning a special interest in structural/visual appearance and time/movement/growth and (2) the ambition to create a 'sculptural moment' i.e. a special interest in forms for the organism to grow on.
I am starting to realise that this might result in the approach that I want to create first a setting/environment/habitat( i.e. sculpture) and then let the organism live, grow, decay on it's own accord. So, creating an interactive setting and then re-treating the human influence. This would include a process of first understanding both the organism's natural (the woods) and man-made (the petri dish) habitat to then create a hospitable setting which will allow the organism to thrive and live autonomously (as it should/could).
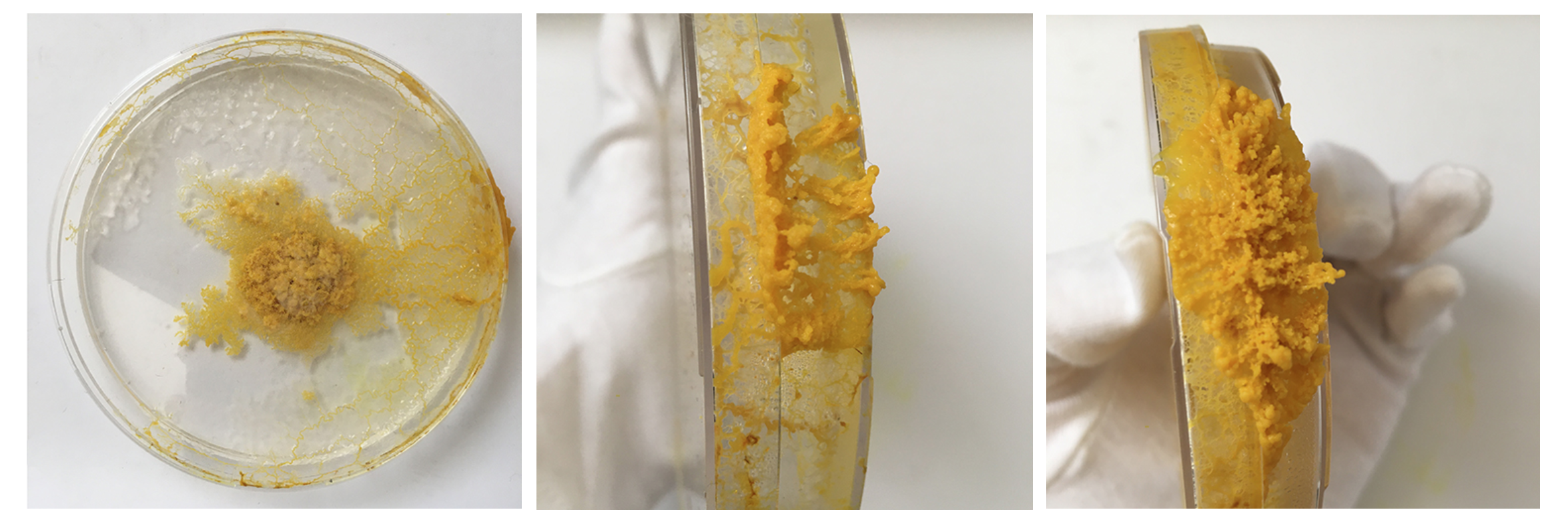
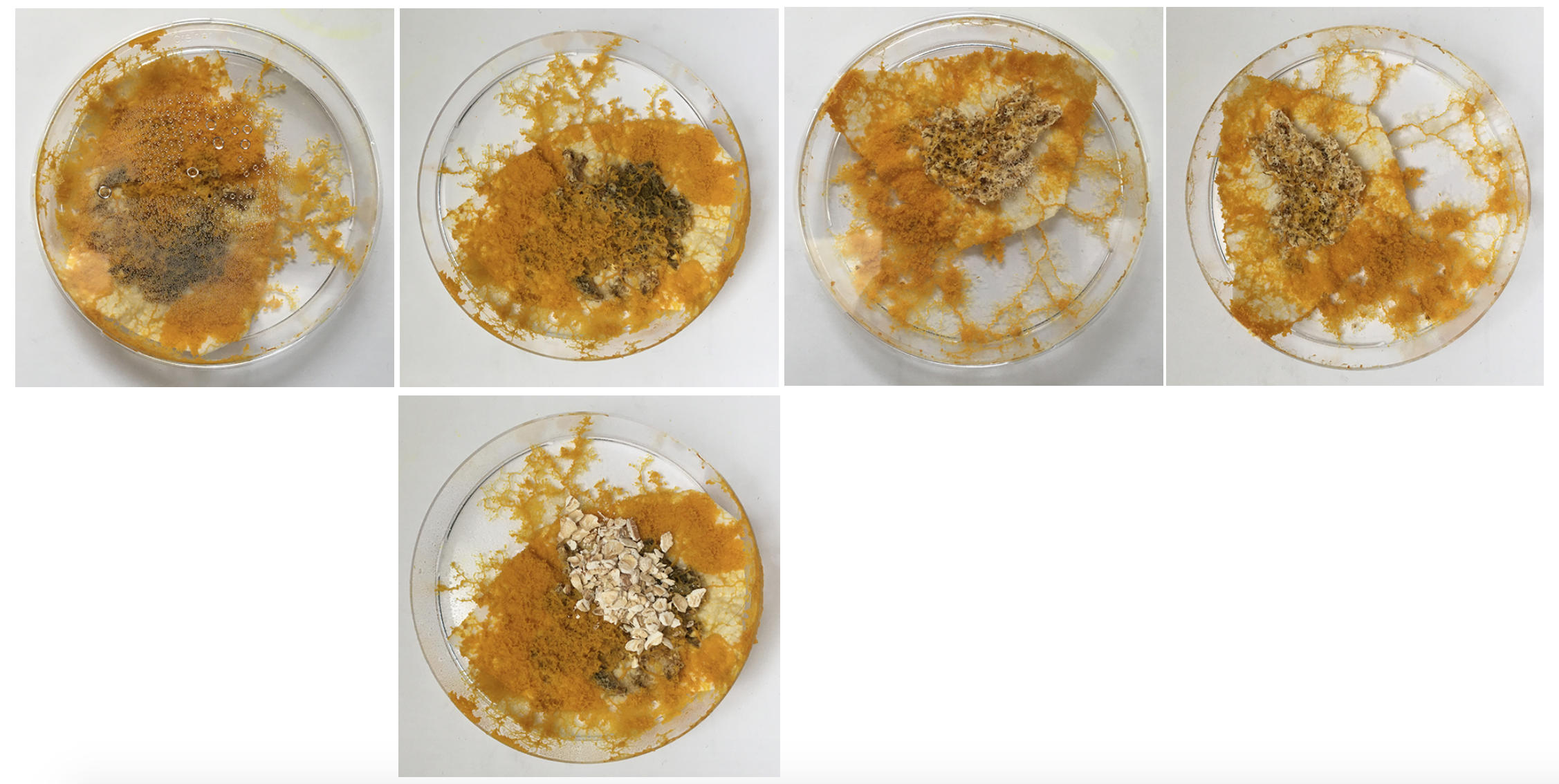
Sun. 31.05 Starting to notice this white-ish slime. First I thought it was mold but now I think it is the trace that Physarum leaves behind itself - which if it has enough space hinders it from going to this spot again. Might be having a strange reaction with the Agar?
Sun. 31.05 Seeing an answer to the question: does physarum grow on surfaces that are not Agar: yes! Does it maybe draw the moisture from the surface of agar so that it doesn't need it in other/all areas?
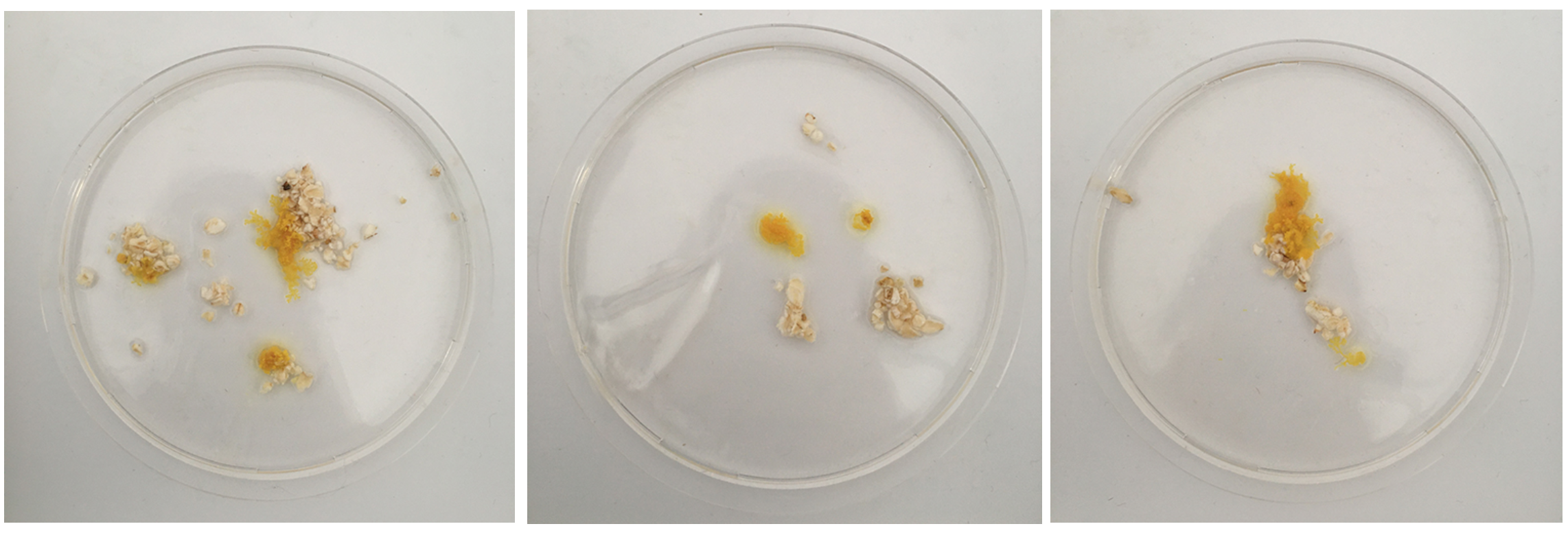
Also you can start to see the digested oats that are no longer yellow so the blob moved off of them because they offer no more for it.
Sat. 30.05 - decided to 'feed' some less to start seeing a difference in behaviour
Sun. 31.05 - because I don't want to much moisture to build up (might lead to bacterial mold) and to keep caring well for the Physarum & first get a better understanding of it, I gave each dish a full spoon of oats today.
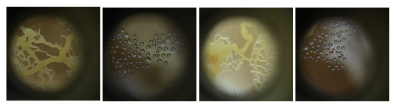
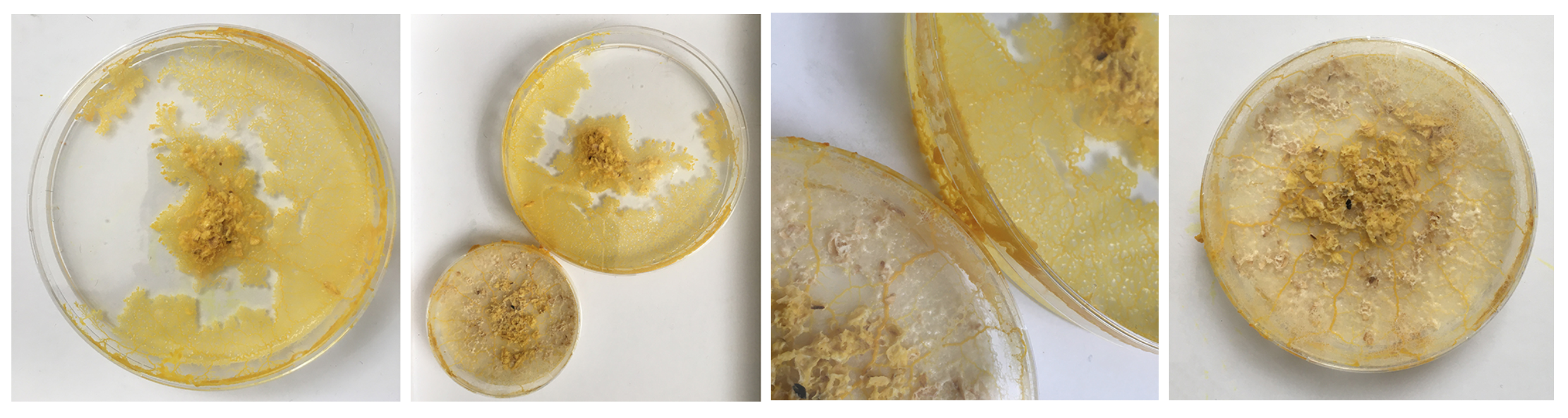
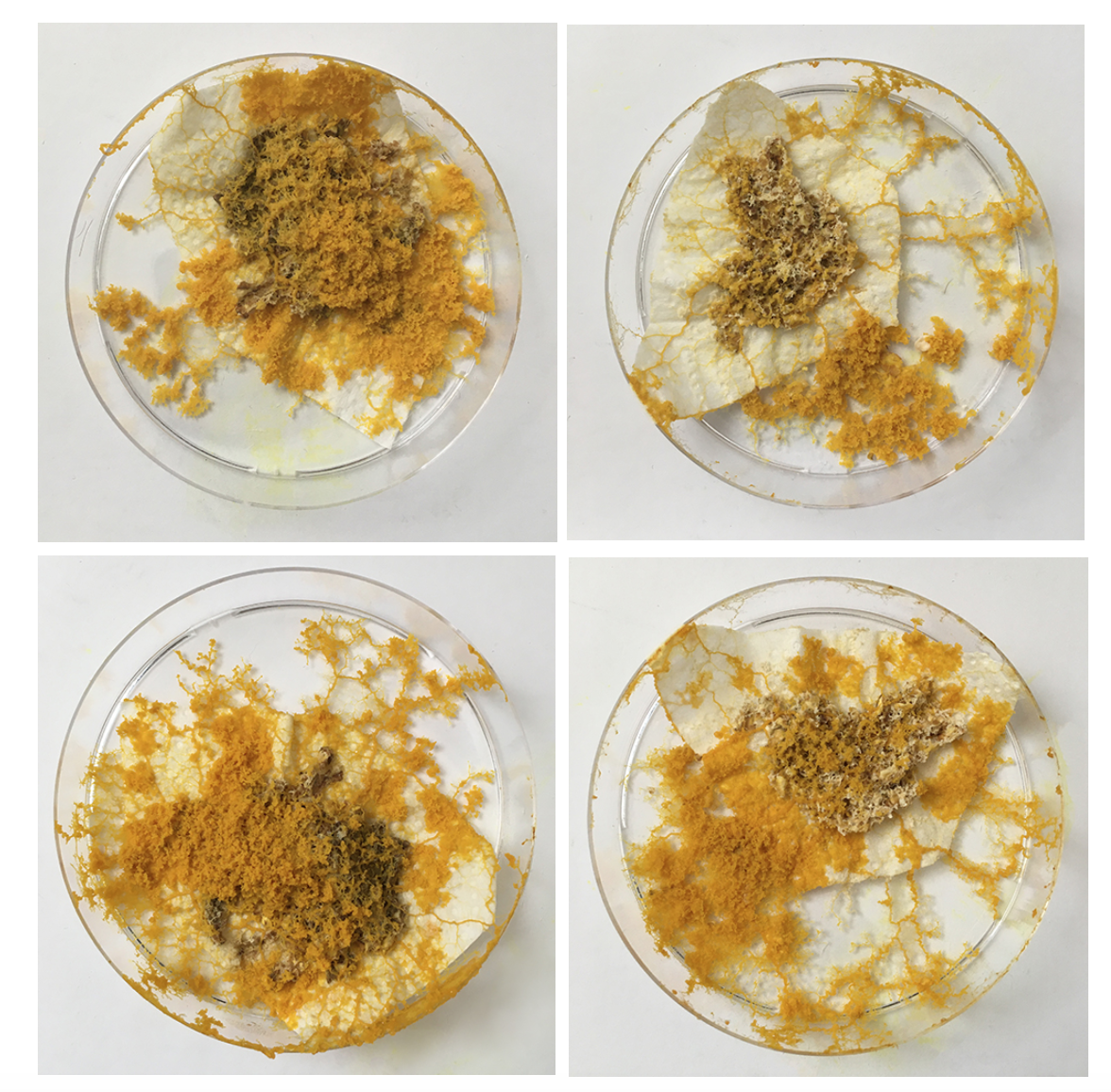
The Physarum's structure is obviously much better visible when NOT ON OATS - so what are aesthetic and sustainable ways to replace the oats or make the food source disappear?
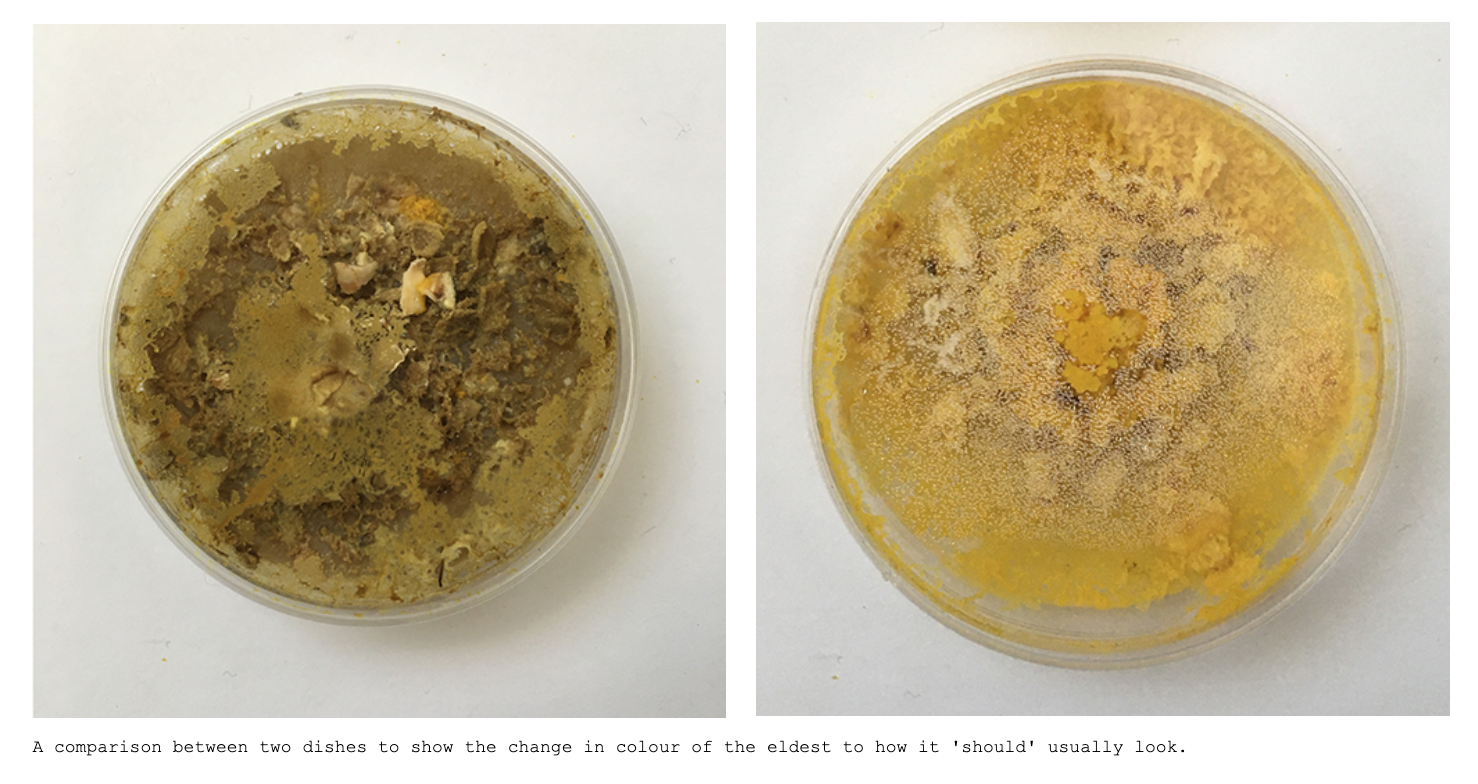
Mon. 01.06 - The first petri-dish, probably older than a week now changed colors, it doesn't look like mold but it's clearly much much darker then if was yesterday. Also doesn't look like the other phases of the life cycle?
Mon. 01.06 - Also there is a lot more moisture in the dishes again - maybe today is finally a day to inoculate? Clearly 6 days is the limit - better to change them after 5 days - like this guide also suggests: https://drive.google.com/drive/folders/1BYdpvPxnUNgdqqu1ELHTnbWlc0cQqvu6
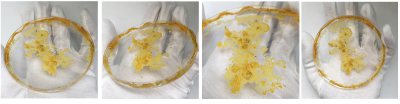
Mon. 01.06 - Here you can see Physarum build a squiggly mass on top of the oats, it's more like a pile of yellow little blobs than the vein like structure.
Mon. 01.06 - Also other dishes are starting to form black dots which I can see more clearly from the bottom - which show bacterial/Yeast contamination and indicate that I should change the dish. Clearly the Physarum wants out, but isen't even healthy enough to make it.
A new inoculation follows!
What isen't really clear to me is what to do with the old dishes from which I inoculate new ones? Do I let them go their course? Do I try to transfer as much of the blob and then let the leftovers dry?
Answer - Mon. 08.06: I wait until the dish is fully dried out our possibly contaminated to throw away whole dish in worst case or clean out the already dried Physarum. I now have pre-made petri-dishes for when a lot tries to escape. If only a little tries to come out I put it back in the same dish.
Observational notes
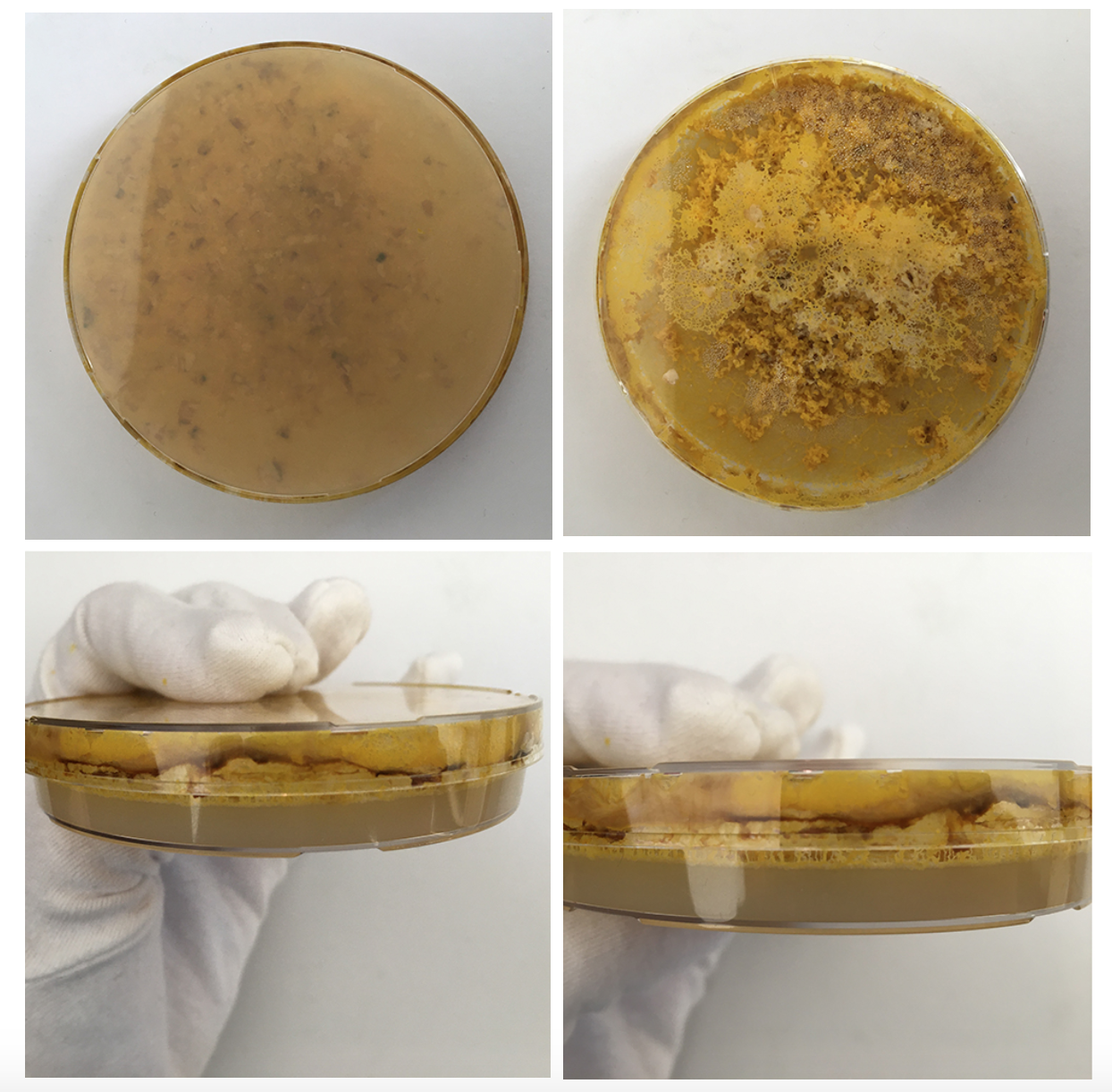
1 large petri dish the agar never settled even though it is the same mix as the others- possible because I moved it too soon disrupting the cooling process?
Also I need to make sure I have a bit more Agar next time as the solution is very wet in general.
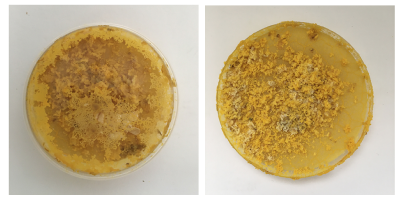
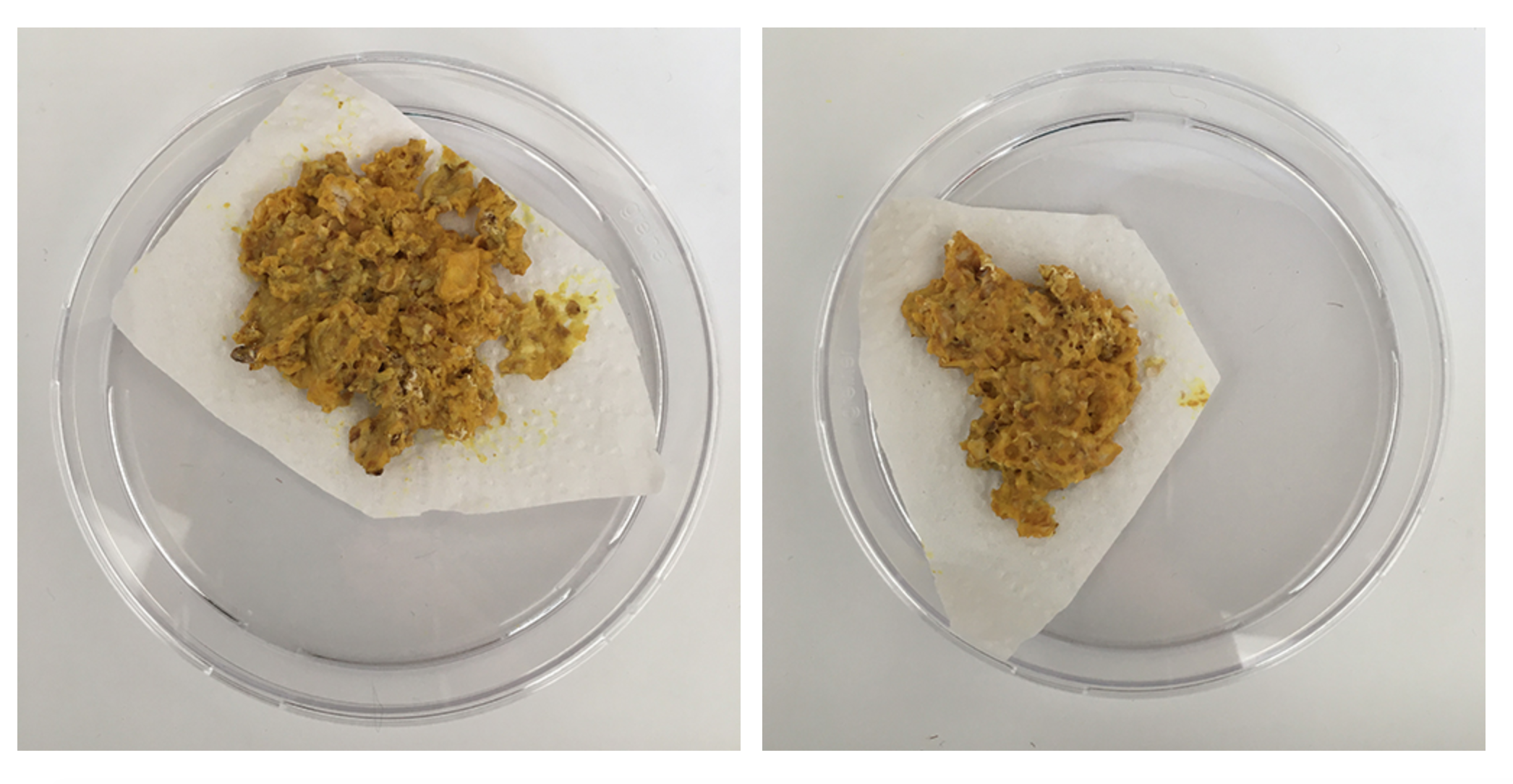
The petri-dishes with the discarded oats and Physarum clearly show that Physarum senses no more food and is trying quickly to escape to survive somewhere else.
Also the medium older dishes show me that Physarum has a sense for the environment and tells me very clearly it wants a new home! Even with enough food it wants to move on somewhere with better conditions - within one night it's climbed half way out the lid. The others are quite happy where they are though.
The small dishes - after I removed the excess /old food seem 'happy' - they've reconnected and are staying inside the dish, also no colour change but the structure looks like it might be going towards spores.
I'm getting a sense for how Physarum perceives it's environment slowly.
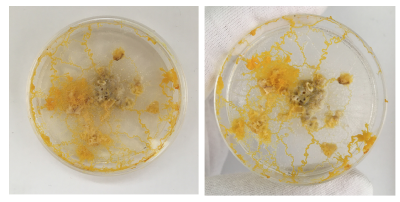
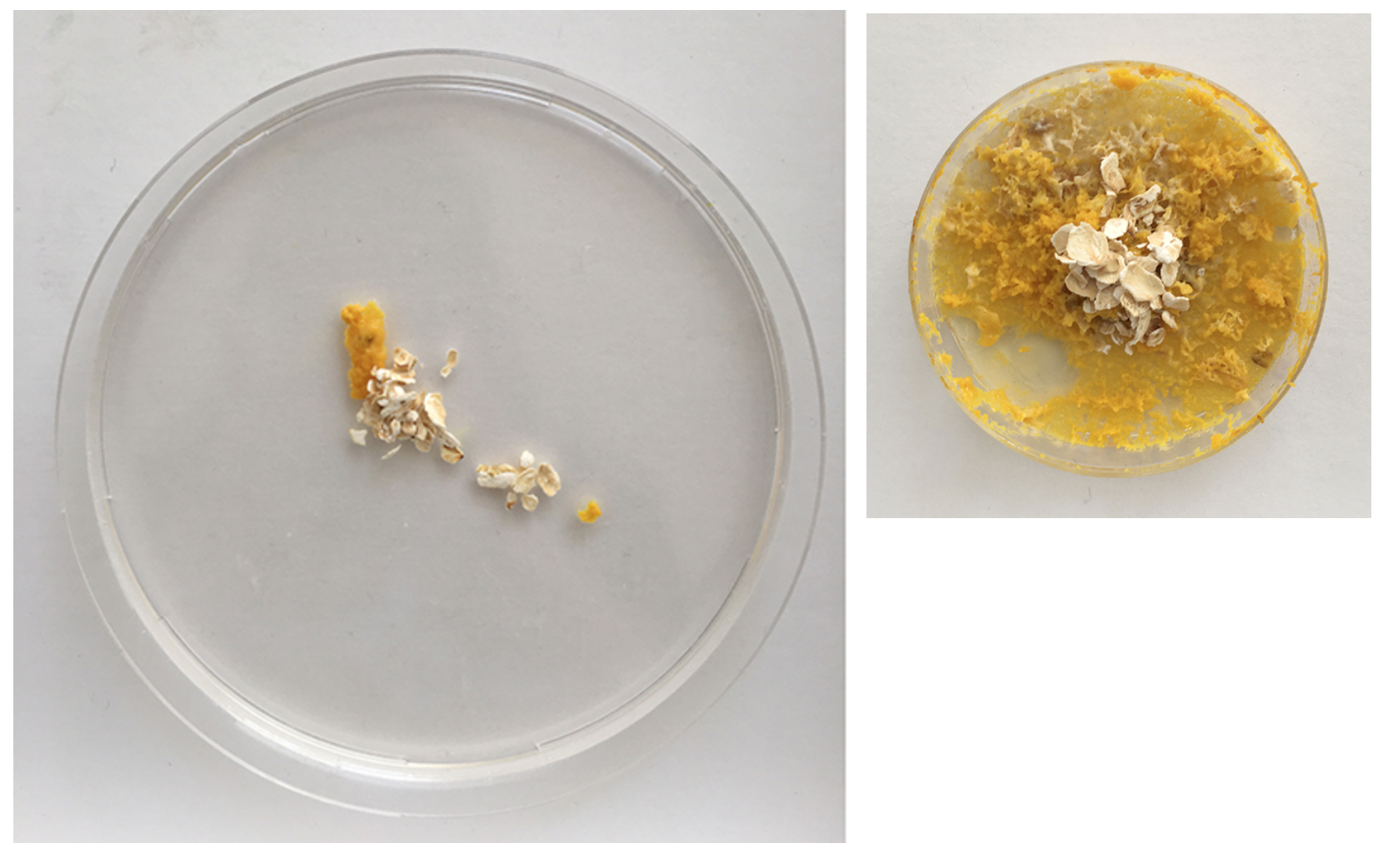
Day 1 - freshly transplanted
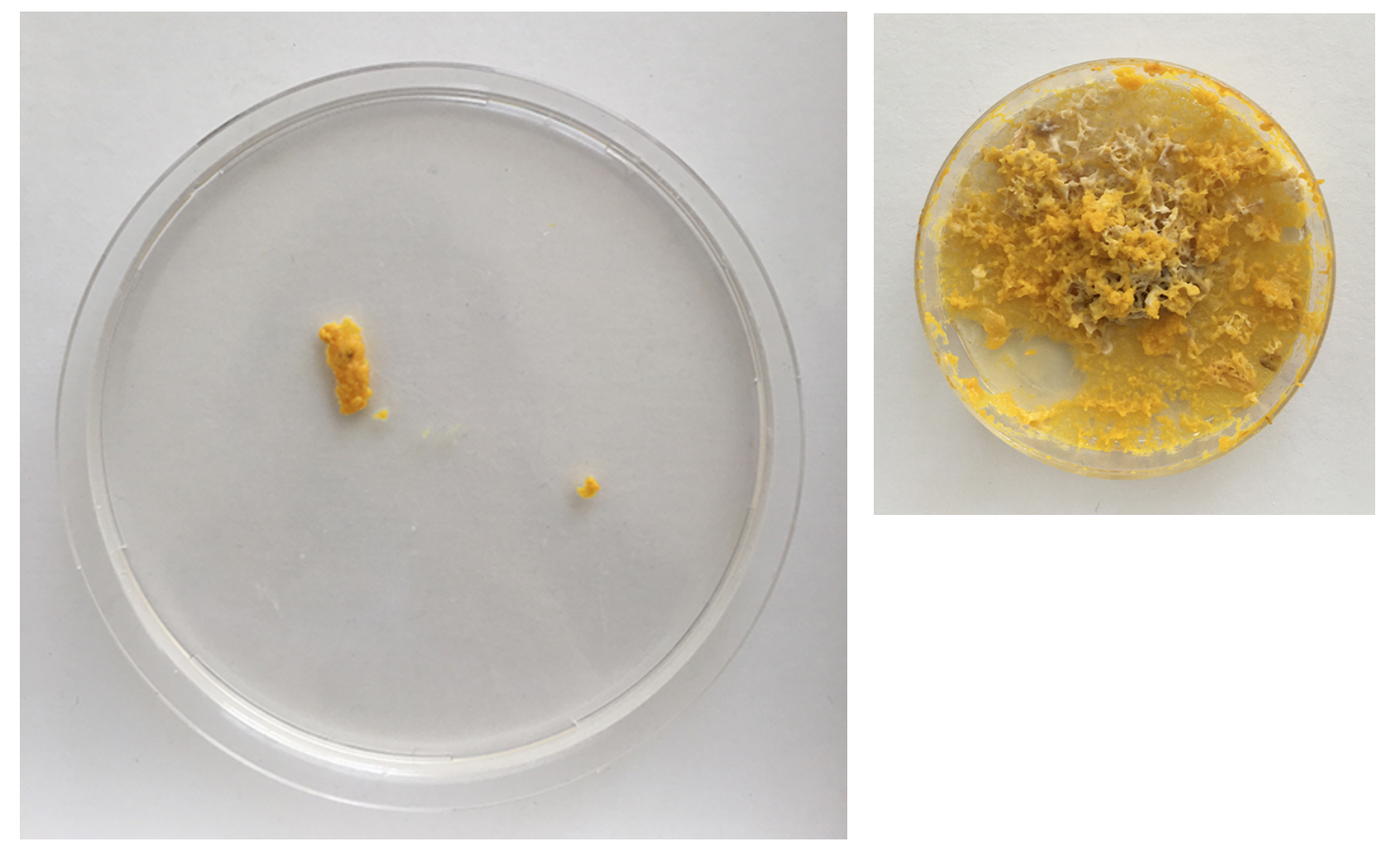
I removed the oats with Physarum from the old dishes and preserved it.
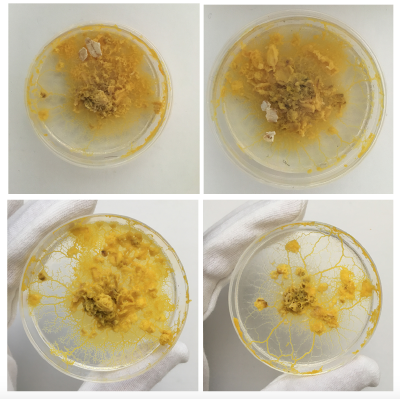
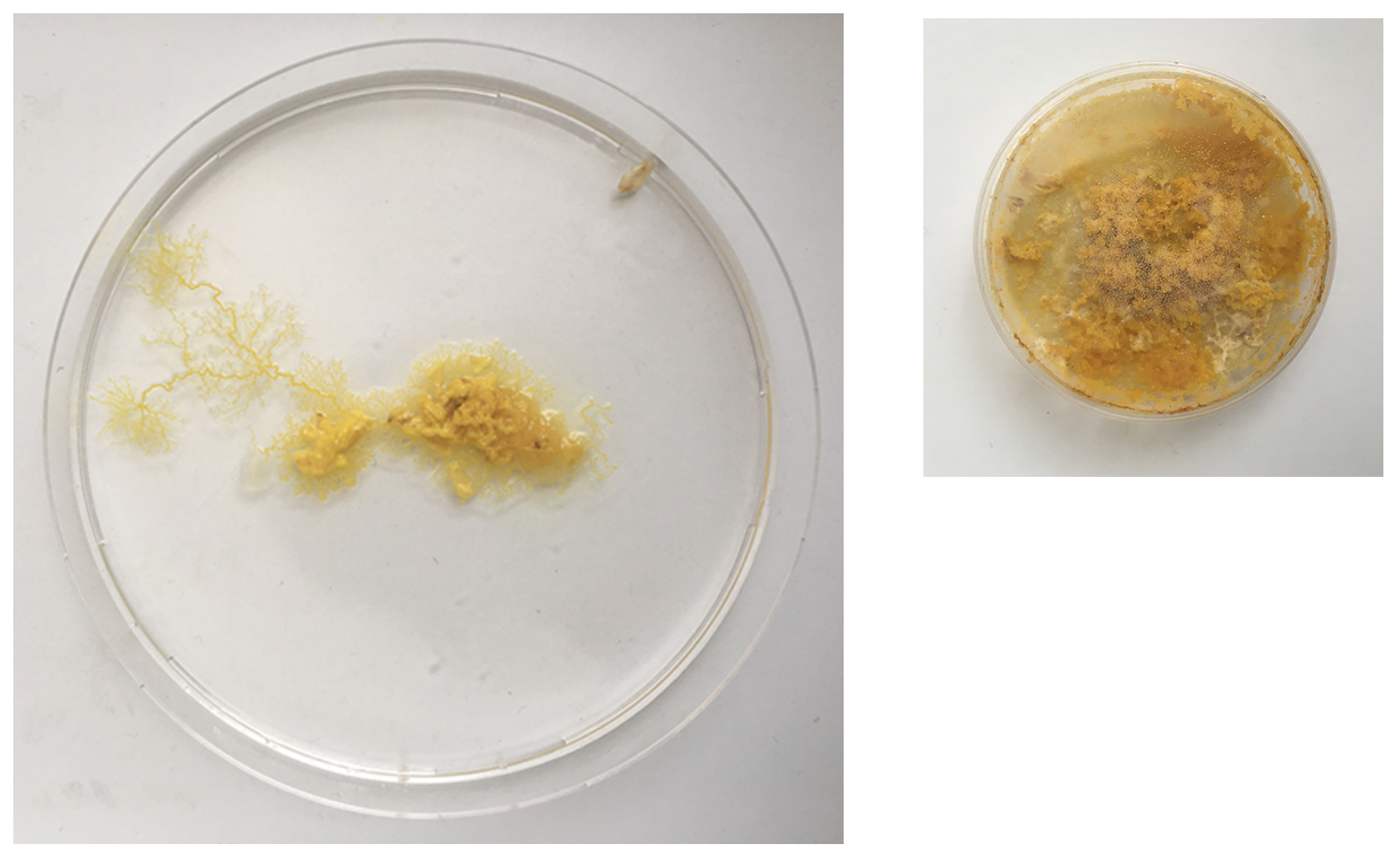
Day 2 - still strong but also fast moving out of the dish. clear indicator that the leftovers can't just be thrown away as the organism still lives on even when perceived as 'discarded' by me. Even spreading faster than on the new Agar dishes.
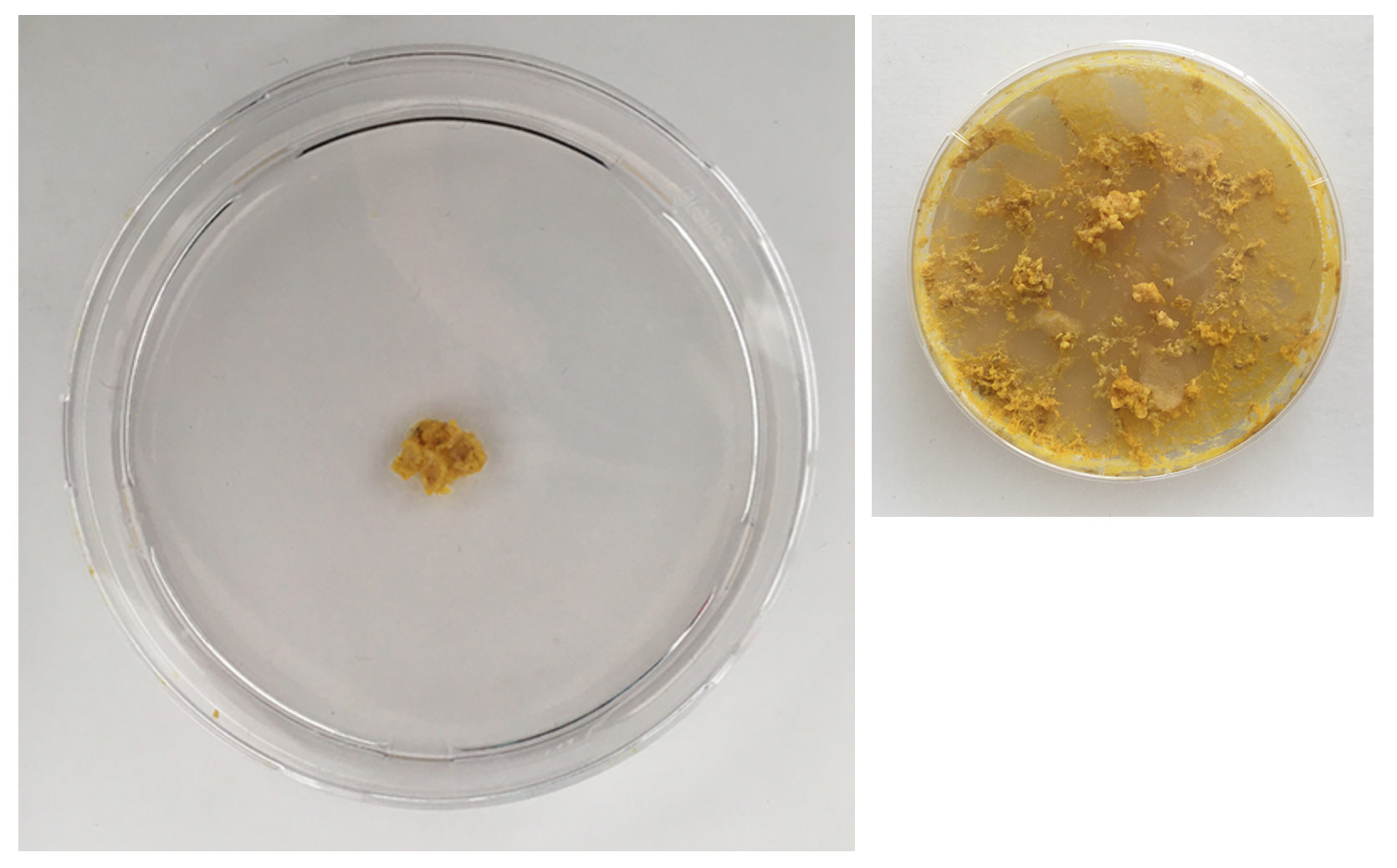
Day 3 - The structure already looks much weaker and the colour is more faint
Day 1 - First movements in the big dishes
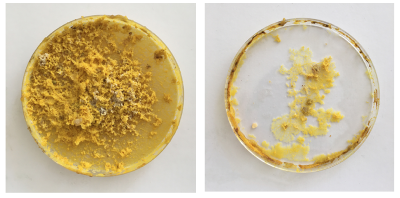
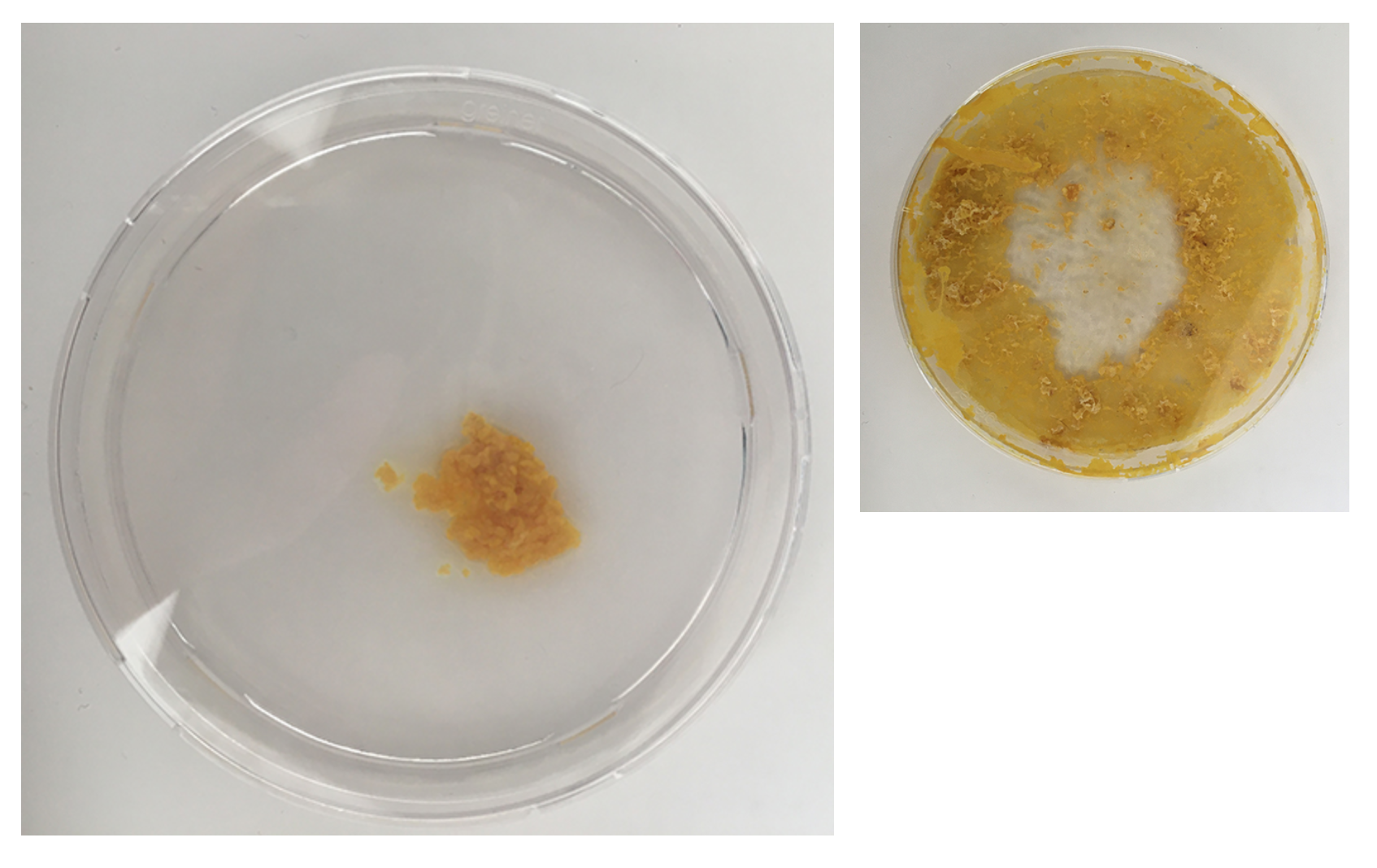
All small dishes are starting to look 'healthier' again - strong structure and colouring
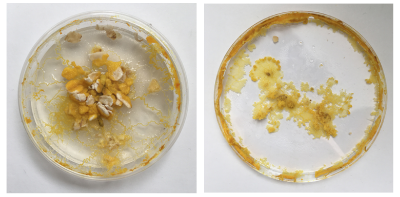
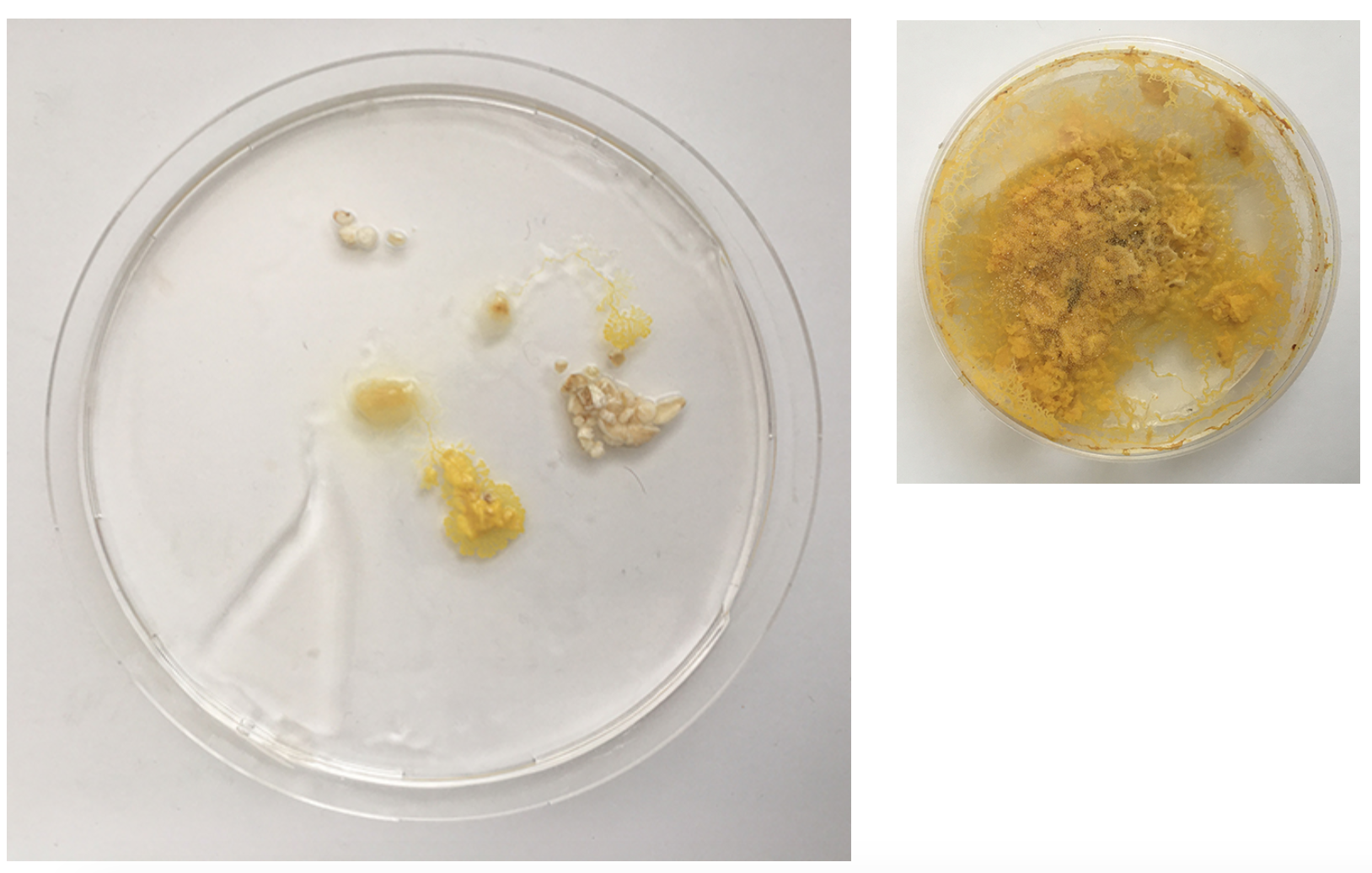
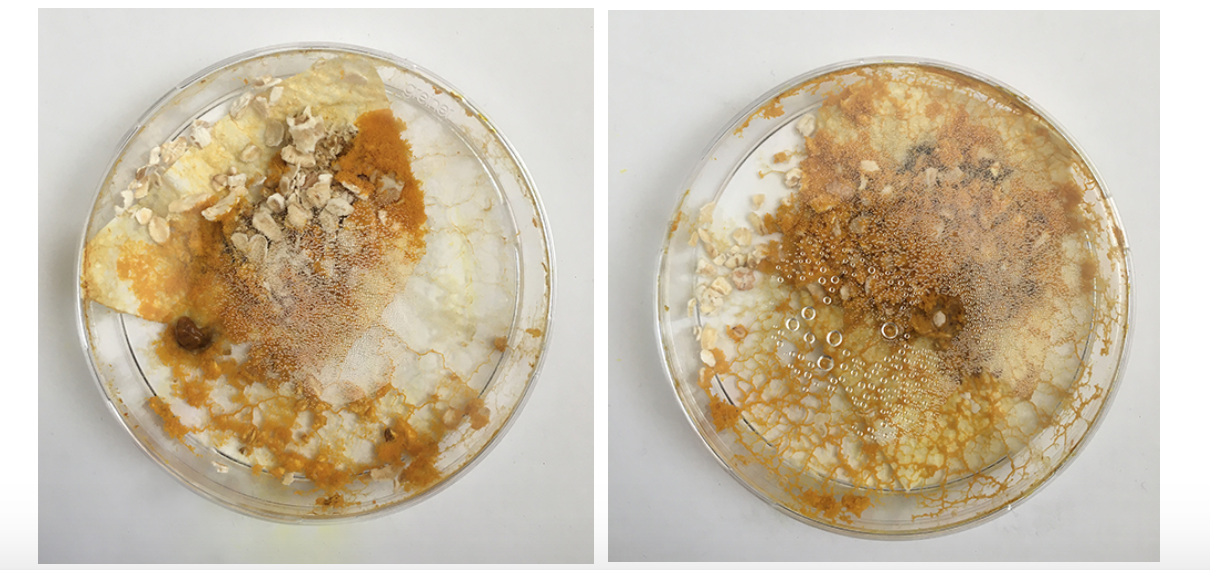
I had these liquidy membrane looking blob parts - what are they? A weird form from the Agar which was too liquid? Answer: When compared to the freshly inoculated dish: these are the spots of the Physarum placed - it moved from this spot quickly on to the fresh oats and these spots might have had a strange reaction with the still too fresh Agar or brought rests that where no longer good for Physarum and turn into this weird blob.
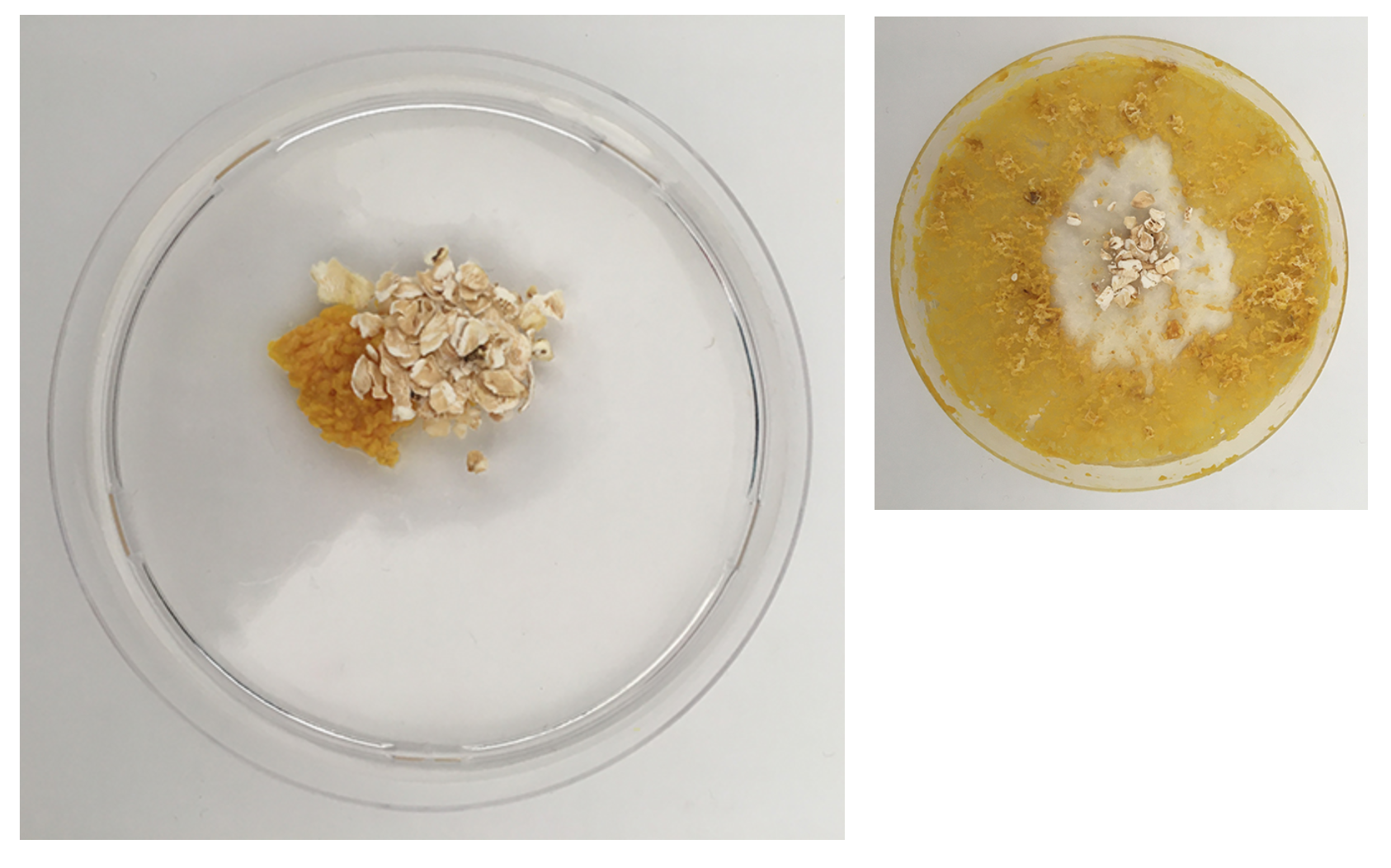
Here, in both dishes, we can see the dish looks quite dry - in the one on the left the Physarum is crawling out because the environment is not good enough anymore.
In comparison the new dishes have a very moist agar (too moist) and are spreading out across the space.
specific documentation of Physarum in captivity: https://kjakubek.hotglue.me/?iforganismdocumentation/
online Processbook in process: https://kjakubek.hotglue.me/?Iforganism